SōRSE News & Blog
Home > News/Blog
More results...
The New Frontier of Infused Products: Powered by Custom Emulsions
Learn how custom emulsions are the new frontier of infused products!

Powered By SōRSE Partner Spotlight: Mad Tasty
Learn how SōRSE worked with Ryan Tedder to develop Mad Tasty!

Water-Soluble Technology: The Solution For Personal Care Products
Learn about the benefits of water-soluble technology for personal care products!

What You Need To Know When Evaluating Water-Soluble CBD Emulsions
Read the 5 tips to evaluating water-soluble emulsions!

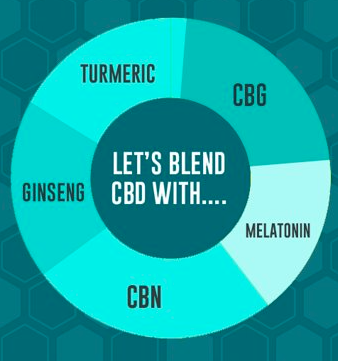
Functional Ingredient Trends: Restoring Protein, Balancing Gut Health, & Boosting Immmunity
Learn more about popular functional ingredients!

Webinar: Cannabis Infused Products: Concept To Commercialization
Check out our co-hosted webinar with New Frontier Data!

5 Steps for Taking Your CBD Edible from Concept to Commercialization
Learn about the key steps in building a CBD edible!
- « Previous
- 1
- 2
- 3
- 4
- Next »









